See also: Atomic Number – Does it conserve in a nuclear reaction? Atomic Number and Chemical PropertiesĮvery solid, liquid, gas, and plasma is composed of neutral or ionized atoms. It is the electrons that are responsible for the chemical bavavior of atoms, and which identify the various chemical elements. In a neutral atom there are as many electrons as protons moving about nucleus. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. The nucleus is composed of protons and neutrons. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. K) - Thermal Conductivity 10 Specific Heat - Heat of Fusion 7.92 Heat of Vaporization - Atomic Number of Berkeliumīerkelium is a chemical element with atomic number 97 which means there are 97 protons and 97 electrons in the atomic structure.Seaborg, Kenneth Street, Jr., Albert Ghiorso Thermal properties Melting Point 990 Boiling Point 2630 Thermal Expansion µm/(m Alkali metals also owe their high reactivity to their large atomic radius - because of this, the lone electron is far from the nucleus, and easily escapes the nucleus’s positive charge.Berkelium – Properties Element Berkelium Atomic Number 97 Symbol Bk Element Category Rare Earth Metal Phase at STP Synthetic Atomic Mass 247 Density at STP 14.78 Electron Configuration 5f9 7s2 Possible Oxidation States +3,4 Electron Affinity - Electronegativity 1.3 1st Ionization Energy 6.23 Year of Discovery 1949 Discoverer Stanley G. This gives the elements a relatively low ionisation energy - they form cations with a charge of +1. Alkali metals have one electron in their outer shell and tend to donate this electron in reactions with nonmetals to form ionic compounds. Alkali metals also have high conductivity and low melting points.Īlkali metals are so reactive due to their electronic configuration. 
For example, lithium sizzles when combined with water, but caesium reacts explosively. These metals increase in reactivity as you progress down the periodic table. They react strongly with water to form hydroxides, and directly with oxygen to produce oxides. Because of this, they are rarely found in elemental form, and are instead most often found in compounds with other elements. They are highly electropositive - meaning they have a tendency to give away their valence electron. The Alkali metals are the most reactive metals in the periodic table. Alkali metals, particularly sodium, are important in commercial use and chemical synthesis.Īlkali metals are highly conductive soft metals, which have a high lustre that oxidizes quickly when they are exposed to air. For example, potassium hydroxide, or lithium hydroxide.Īlkali metals have been studied since 1807, when Sir Humphry Davy explored the electrical properties of potassium and sodium. They are called alkali metals because they react strongly with water to form alkalies - hydroxide compounds made up of the element plus an -OH (hydroxide). Why are the group one elements called alkali metals? The alkali metals are located in the first column of the periodic table and include all the elements in that column except for hydrogen.ģ. Where are the alkali metals located on the periodic table? Thus, this electron can easily escape the positive pull of the nucleus to be donated to other elements, resulting in a reaction.Ģ. FAQsĪlkali metals are so reactive due to their electronic configuration - they have one valence electron and a large atomic radius. 
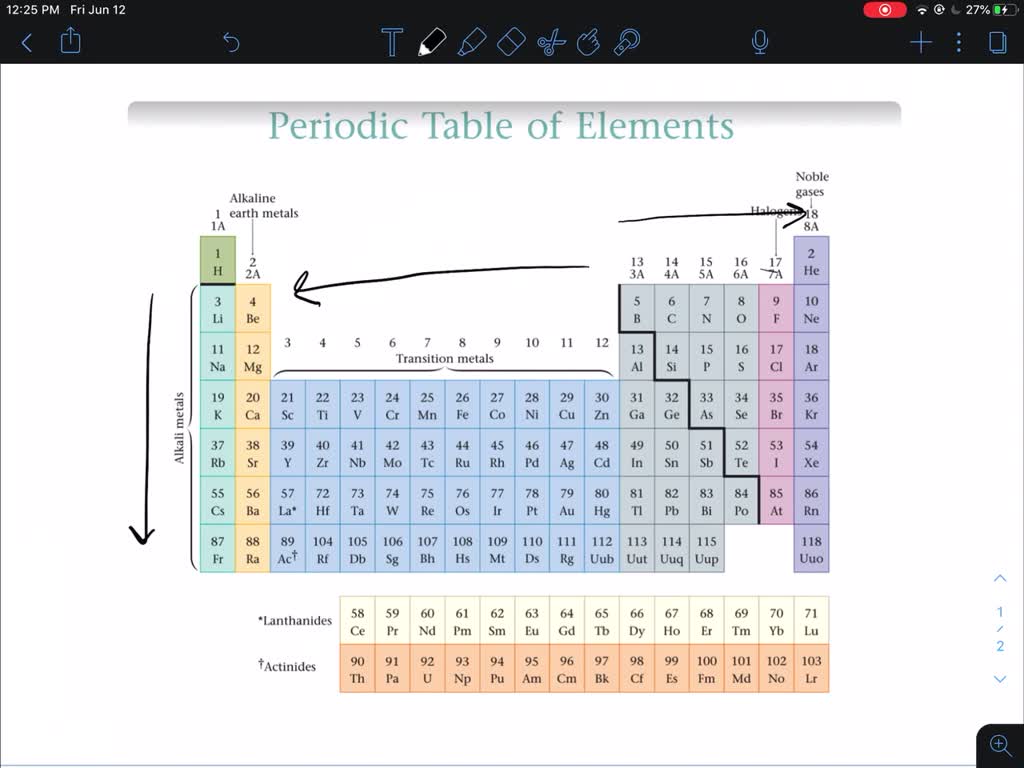
Metals make up almost the entire left side of the periodic table, save for Hydrogen which is a nonmetal. Elements in the periodic table can be described as metals, metalloids, and nonmetals. Elements in the same group typically have similar chemical properties as a result of their similar electronic configuration. All of the elements in a group share the same number of valence electrons: electrons in their outermost shell. Refresher: The periodic table is organized into groups - where each column comprises a group.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
